Glycolysis occurs in the cytosol, while the oxidative decarboxylation of pyruvate occurs in the mitochondria, and the citric acid cycle occurs in the mitochondria.
NAD+ is in E3
Coenzyme-A is in E2
Lipoate is in E2
FAD is in E3
TPP is in E1
The key is that pyruvate to acetyl-CoA is irreversible, so there is no way we can get back pyruvate or sugar.
Incorporation into fatty acids
Complete oxidation to form carbon dioxide.
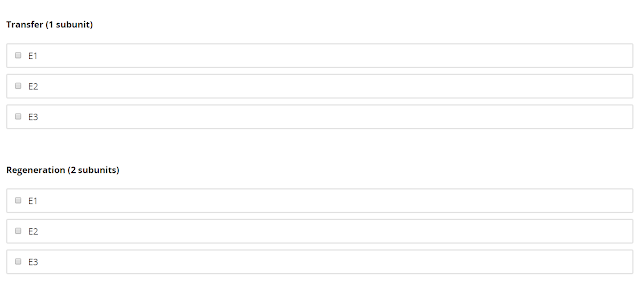
This question is "tricky" - there is more than 1 unit for certain steps, it is clear in the problem but I still missed it.
Decarboxylation happens in E1, where pyruvate attaches to TPP.
Oxidation happens in E1 and E2, where the molecule linked to lipoamide with a thioester bond.
Transfer happens in E2, where the molecule is now attached to CoA instead.
Regeneration happens in E2 and E3, the lipoamide now forms the disulfide bond.
PDH is allosterically activated by its substrates and
PDH is allosterically inhibited by its product.
This is simply the Le Chatelier's principle.
The pyrudehydrogenase kinase phosphorylate PDH, so when pyrudehydrogenase is activated, the PDH is deactivated and we generate less energy.
NADH ATP and Acetyl-CoA are products of the PDH, so they should deactivate PDH.
Pyruvate is product of PDH, so it should inhibit PDH.
At low energy state, ADP accumulates, to it should inhibit PDH
Calcium does not impact PDH.
PDH is the gateway into the citric acid cycle, so it is tightly regulated.
Thioester bond cannot be stabilized by resonance structure, therefore it is less stable and has a high free energy of hydrolysis. We need to have this high free energy state to form Acetyl-CoA, which also have a thioester bond.